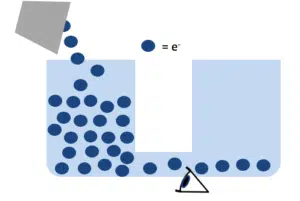
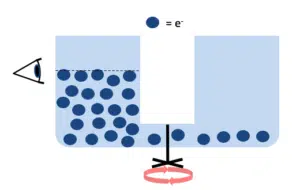
Galvanostat
If you want to apply a current to a liquid, such as mercury, and want to observe the change of potential, you need a device that can control the former and measure the latter. Such a device is called a galvanostat.
Luigi Galvani
The word galvanostat comes from an Italian scholar named Luigi Galvani (1737-1798). He discovered that a difference in electronic charge (potential) causes motion. His work is known as the basis for the invention of the battery.
How does a galvanostat work?
A galvanostat is capable of increasing or decreasing a potential over two points in a solution by applying a stable electron current. When there is a difference in the potential between the galvanostat and the liquid, the potential will want to level itself, which will cause the potential in the liquid to drop or increase. The measurement shows how this effects the potential of the liquid (how quickly the potential increases/decreases in time).
Galvanostat applications
Galvanostats are sometimes applied as battery chargers. Researchers want to know how long a particular battery will last. They will test this by constantly charging and de-charging the battery and monitor the characteristics. Also a battery charger that consumers use at home is a kind of galvanostat.
Another application is when the resilience of a certain protective layer needs to be determined. By applying an electron current on the layer, you can measure how high the potential of the layer is and how it will behave under different circumstances.
Galvanostat software
Without software, a galvanostat is of no use. With our dedicated software you can control the instrument, apply the experimental settings and see the measurement data in many graphical representations. Our software always includes free use on multiple computers and free updates. The PalmSens4 galvanostat is sold including its own easy-to-use intuitive software, which allows you to execute your experiments immediately without any required training. If you prefer you can also use all common spreadsheet software for creating your own graphs with the exporting functions available in our PSTrace software.
Galvanostat prices
A high quality galvanostat can be expensive. Potentiostats are available from hundreds of euro or dollar to tens of thousand euro or dollar. PalmSens managed to develop a galvanostat, the PalmSens4, that is highly affordable, with excellent instrument specifications.
You can try for yourself and compare our specifications with specifications of competitors. If you are interested in a specific instrument, please add the instrument to your shopping basked and request a quotation. We will get back to you within one business day. Feel free to call us to discuss any possibility for your research.
Select an instrument and request a quote- Galvanostat
- A device with which you can apply a constant current to for instance a liquid such as mercury and observe the change of potential