Compliance voltage
The compliance voltage is the maximum voltage that can be applied between the working and counter electrode. Another name could be the maximum cell potential.
Purpose of a Potentiostat
The purpose of a potentiostat is to apply a known potential difference between the working electrode and a known reference point, which is the reference electrode. To keep the reference electrode’s potential stable the current should not flow through the reference electrode.
A third electrode, the counter electrode, carries the current load. This means a defined potential is applied between the working electrode and the reference electrode, but the current is measured between the working and the counter electrode.
Cell potential for current flow
For a current to flow a potential must be applied between the working and counter electrode. This is the cell potential. It is adjusted in such a way that the potential difference between the working electrode and the reference electrode is the set potential. Or in other words, the counter electrode’s potential is adjusted in such a way that sufficient current flows between the working and counter electrode.
The flowing currents can be caused by the charging of the electrochemical double layer (capacitive charging current) or an electrochemical reaction (Faraday current). The current at the counter electrode and the working electrode must have the same amount but opposing polarities, for example, if -1 mA flows at the working electrode +1 mA must flow at the counter electrode.
If small currents are flowing at the working electrode and a large counter electrode is used the capacitive charging current can suffice as the counter electrode’s current. In most situations, an electrochemical reaction will take place at the counter electrode.
Oxidation and reduction
Oxidation at the working electrode requires a reduction at the counter electrode and vice versa. The oxidation and reduction occurring at the counter and working electrode define the minimum cell potential required for the cell. The potential difference between the working electrode and the counter electrode needs to be big enough to drive both reactions.
Often the design of an experiment does not take the reaction at the counter electrode into consideration. For that reason, the most common reaction at the counter electrode is the splitting of the solvent, which is for example water, i.e. the oxidation of water to protons and oxygen or the reduction of water to hydrogen and hydroxide.
For example, the oxidation of iron at the working electrode occurs at +200 mV vs RE. Water splitting is the only possible reduction at the counter electrode. The reduction of water occurs at -1.2 V vs RE. This means the cell potential, the difference between the working electrode and the counter electrode is -1.4 V at least.
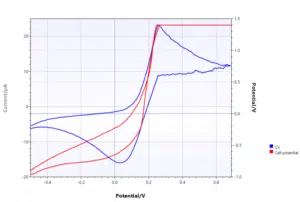
Maximum cell potential reached
The higher the current at the working electrode, the higher the current required at the counter electrode. To increase the current the potentiostat increases the potential difference between the working and counter electrode. At some point, the maximum cell potential is reached and cannot be increased further.
This means the working electrode’s potential might not be correct anymore, because the correct current cannot flow through the counter electrode anymore. The potentiostat ran into its compliance voltage. The potential of the working electrode will stay at the last value it could reach.
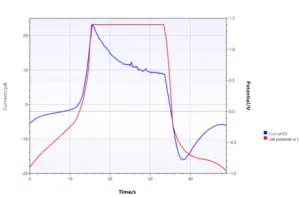
During DPV or SWV this means the differential is 0 and thus the curve shows a sudden drop in current and stays at 0. In a Cyclic Voltammogram reaching the compliance voltage is clearly visible. The constant potential will lead to a current following the Cottrell equation:
The plot of current versus time clearly shows that the current shows a time dependence as described by the Cottrell equation instead of the expected potential dependence.
Curious about the compliance voltage of PalmSens potentiostats? Select the potentiostats of your interest on the product comparison website.
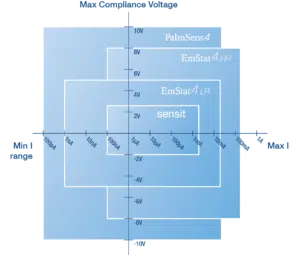
Compliance voltage per instrument
The compliance voltage differs per instrument. In this overview you can compare the instruments.
Note that the EmStat Pico module has the same compliance voltage as the Sensit Series potentiostats.