Salt bridge
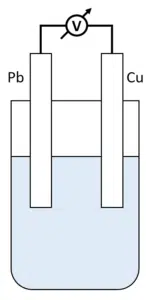
When two electrodes are in the same solution, and a potential is applied, ions flow from one electrode to the other through the solution. This forms a layer on the electrodes, leading to unstable measurements.
A salt bridge allows the current to flow through it, whilst maintaining electrical neutrality.
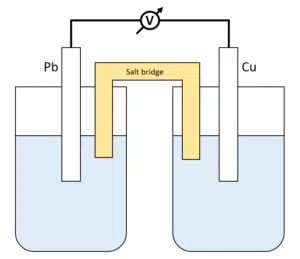
Salt bridges are often made of filter paper or, the more durable option, (glass) tubes.
Articles

Electrodes used with a potentiostat
This section gives a short overview of the three types of electrodes (working, reference, counter electrode) you will encounter while using a potentiostat. It is explained how these electrodes look like and what their task is.
