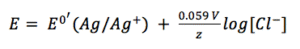Reference electrode
The reference electrode should deliver a constant potential. A current flowing through an electrode leads to an electrochemical reaction that will change the composition of the electrode’s environment and thus the potential. As a consequence there should be as little current as possible flowing through the reference electrode.
To establish and keep a constant potential an electrode of the second type is often chosen. Their potential usually depends indirectly on the concentration of a single anion. The electrodes of the first type are basically just metal surfaces in an electrolyte.
According to the simplified Nernst equation (see equation 3.6) the potential is depending directly on the surrounding solution. An electrode of the second kind is usually a metal surrounded by a hardly soluble salt of itself. This electrode is then immersed into a solution containing the anion of that salt.
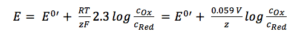
Usually the anion has a high concentration to make sure that the metal salt is not dissolving fast and that small changes in the concentration have a low impact on the potential. A common reference electrode is the silver/silver chloride electrode (see Figure 3.5). A silver wire is coated with a film of silver chloride. This wire is immersed in a potassium chloride solution.
The potential of the silver wire depends on the silver ions dissolved in the potassium chloride solution.
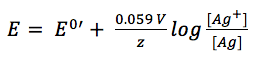
Due to the fact that it is a solid material the concentration of silver is 1 M. The concentration of the silver ions depends on the concentration of chloride, as the solubility product KL (equation 3.8) shows.
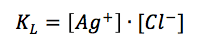
If 3.7 and 3.8 are combined and all constants are summarized in E0’(Ag/Ag+) the result is:
If the chloride concentration is kept constant a good reference electrode is created. This is usually achieved by separating the measuring solution from the solution surrounding the reference electrode using a porous frit.
Even small concentration changes due to diffusion through the frit or evaporation have little impact on the concentration if the concentration of chloride is high. The frit also prevents silver ions from diffusing into the measuring solution. The silver/silver chloride electrode is one of the two most popular chloride-containing reference electrodes.
The reference electrode keeps the system stable. Fluctuations in the potential result in noisy measurements. The reference electrode should be checked first if the system behaves in an unexpected way.
Articles

Screen Printed and Film Electrodes
Screen printed and film electrodes are suitable for mass production, versatile, cheap, and easy to handle. They make electrochemistry available to users without electrochemical knowledge.
Constant Potential vs OCP or vs Reference Electrode
In this section the reasons for choosing a certain DC potential is explained. The advantages of using Ecorr as a reference point will be presented as well as how to choose in PSTrace to use potentials versus the reference electrode or versus Ecorr.

Electrodes used with a potentiostat
This section gives a short overview of the three types of electrodes (working, reference, counter electrode) you will encounter while using a potentiostat. It is explained how these electrodes look like and what their task is.