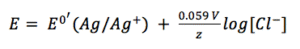Electrodes used with a potentiostat
This section gives a short overview of the three types of electrodes (working, reference, counter electrode) you will encounter while using a potentiostat. An explanation is given of what these electrodes look like and what their task is.
Electrodes in Electrochemistry
The potentiostat is at the heart of an electrochemical experiment and modern potentiostats need little effort to set up and operate. The average electrochemist will spend a lot more time with their electrodes. They need to be polished maybe modified, and sometimes it is rather challenging to fix them all in the right place.
For this reason we will first talk about the three different electrodes, before we take a closer look at the potentiostat again.
Working Electrode
The working electrode is the place where the reaction happens that we want to control or investigate. As a consequence, this electrode should be carefully and reproducibly prepared. The most common working electrodes are disc electrodes.
For example, a metal cylinder or a metal wire is surrounded by Teflon or PEEK and the cross-section is exposed. The metal disc is connected to a wire at the other end of the coating, so it can be connected (see Figure 3.2). Common materials in electrochemistry for working electrodes are platinum and gold as well as a variety of carbon phases. Very popular due to its conductivity and reusability is glassy carbon.
In corrosion research, the working electrode is usually the surface or material that is to be studied. So in corrosion research, most cells or sample holders focus on exposing a defined area to the solution in which the measurement is performed as well as providing an electrical connection of the sample (see Figure 3.3).

- Working electrode
- The working electrode is the electrode where the investigated processes occur. It needs to be carefully prepared, so that the surface is reproducible and known.
Stern-Makrides arrangement
Another option to set up your sample is the Stern-Makrides arrangement. The sample should have a cylinder shape and a hole with a screw thread. A Teflon cone is used to make the connection to the metal rod watertight (Figure 3.4). There are many other cells that allow insulating different parts of samples. There are cells for metal sheets or cells that are similar to a scanning droplet cell, where an electrolyte-filled tip is pressed to the surface and turns thus the part in contact with the liquid into a working electrode.
Rough surfaces and cleaning
Rough surfaces will lead to high currents due to the charging of the interface, which will be explained in the chapter Capacitive Current. Dirty surfaces can show artifacts in the measurements or can cause the active surface to be only partially in contact with the electrolyte. So cleaning an electrode is an important step in electrode preparation. A clean surface also increases the reproducibility of your measurements. It is therefore recommended to wear disposable gloves when assembling the samples and the holder.

Reference Electrode
The reference electrode should deliver a constant potential. A current flowing through an electrode leads to an electrochemical reaction that will change the composition of the electrode’s environment and thus the potential. As a consequence there should be as little current as possible flowing through the reference electrode.
To establish and keep a constant potential an electrode of the second type is often chosen. An electrode of the second type their potential usually depends indirectly on the concentration of a single anion. The electrodes of the first type are basically just metal surfaces in an electrolyte.
According to the simplified Nernst equation (see equation 3.6) the potential depends directly on the surrounding solution. An electrode of the second kind is usually a metal surrounded by a hardly soluble salt of itself. This electrode is then immersed into a solution containing the anion of that salt.
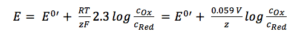
Usually the anion has a high concentration to make sure that the metal salt is not dissolving fast and that small changes in the concentration have a low impact on the potential.
- Reference electrode
- The reference electrode keeps the system stable. Fluctuations in the potential result in noisy measurements. The reference electrode should be checked first if the system behaves in an unexpected way.
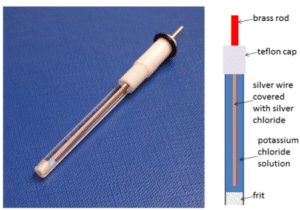
Silver/Silver chloride electrode
A common reference electrode is the silver/silver chloride electrode (see Figure 3.5). A silver wire is coated with a film of silver chloride. This wire is immersed in a potassium chloride solution.
The potential of the silver wire depends on the silver ions dissolved in the potassium chloride solution.
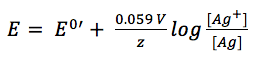
Due to the fact that it is a solid material the concentration of silver is 1 M. The concentration of the silver ions depends on the concentration of chloride, as the solubility product KL (equation 3.8) shows.
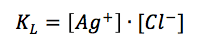
If 3.7 and 3.8 are combined and all constants are summarized in E0’(Ag/Ag+) the result is:
If the chloride concentration is kept constant a good reference electrode is created. This is usually achieved by separating the measuring solution from the solution surrounding the reference electrode using a porous frit.
Even small concentration changes due to diffusion through the frit or evaporation have little impact on the concentration if the concentration of chloride is high. The frit also prevents silver ions from diffusing into the measuring solution. The silver/silver chloride electrode is one of the two most popular chloride-containing reference electrodes.
Other reference electrodes
Another example of an electrode is the calomel electrode. This reference electrode contains mercury in contact with mercury(II) chloride in a chloride-containing solution often saturated with chloride. This is a quite common reference electrode for corrosion cells.
The saturation of the solution means, that solid potassium chloride is inside the glass body. If the electrode’s solution is losing chloride, some of the solid chloride will go into the solution and keep the concentration constant.
You should always indicate clearly versus which reference electrode the potentials were applied or measured, if you publish results or want to compare them to other measurements. Often you find this information in the axis label e.g. “E / mV vs Ag/AgCl” or “E / mV vs SCE”(SCE = Saturated Calomel Electrode).
If the currents are small enough, that is in the nA range, it is possible to immerse the wire with the hardly soluble salt coating in the measurement solution. This is called a pseudo reference electrode.
The reference and counter electrode can also form a short circuit and you can work with a two-electrode system. If your currents are higher, you want to perform a long-term experiment, for example overnight, or need a very stable potential, a three-electrode system with a proper reference electrode is necessary.
Positioning of the reference electrode is often not important, if you have a well-conducting solution. If you have a solution with low conductivity, you might experience Ohmic drop. The Ohmic drop is the amount of potential that is lost on the way from the reference electrode to the working electrodes.
Since this potential is lost, it isn’t felt by the working electrode. This means, if you apply 1 V between the reference and working electrode and have 200 mV of Ohmic drop, your working electrode will behave as if the driving force is 800 mV and not 1 V.
The Ohmic drop is a result of the Ohmic resistance between the reference electrode and working electrode. The resistance depends on the distance between the electrodes, the conductivity of the solution and the frits used. The frits are fixed and can’t be influenced by the user.
The conductivity of the solution is often dictated by the experiments. In a solution of 100 mM KCl is the conductivity sufficiently high, that for most purposes the Ohmic drop is negligible. Corrosion studies sometimes require that the solution is chosen according to the real environment the sample will be exposed to. This could mean a low conducting solution. Here Ohmic Drop could be important.
The distance between the reference electrode and the working electrode is in many setups variable. Unfortunately, a reference electrode that is very close to the working electrode might block the diffusion of species to or from the electrode, creating an artificial crevice. Furthermore, the salt concentration in the reference electrodes body is usually quite high.
Since a frit is not 100 % tight, a bit of the solution in the electrode’s body will leak into the measurement solution. This will lead to increased concentrations of ions in front of the reference electrode.
To avoid all this a Luggin capillary or salt bridge is used. A Lugging capillary is a glass body where the reference electrode is entered into one end and the other end is close to the working electrode. The capillary’s narrow opening limits diffusion, but not as much as a frit.
As a result the resistance is lower than the one of a frit, but if the narrow part of the frit increases the resistance again. Another advantage of the capillary is its small size. The capillary can be approached close to the working electrode with very little disturbance of the diffusion.
Another option is to use a salt bridge, which is what is delivered with our corrosion cell. It allows a close positioning to the working electrode, it has a frit, and is filled with the cell solution.
Counter Electrode
The counter electrode is just some inert metal or carbon species with a large surface. The counter electrode usually needs the least maintenance. For people who work with many different materials, platinum is a good counter electrode due to its inertness towards most solutions. A really big surface area is achieved by a mesh structure, but often a platinum wire has already a surface area several magnitudes bigger than the working electrode. Platinum is also very easy to clean. A hand torch is usually enough for making the platinum glow and to remove all stains.
The counter electrode should have a large surface area, to enable a high current flow even with low potentials. This way water splitting, gas production, or the production of aggressive radicals is avoided. If this kind of reaction is happening in a small volume of solution, it will no longer be negligible. A large surface area will lead to high capacitive currents (see Capacitive Current). At the counter electrode, capacitive currents provide a current flow without changing the chemical composition of the solution.
- Counter electrode
- The counter electrode should not limit the current or influence the working electrode. Popular electrodes are platinum wire or platinum mesh electrodes.