Electrochemical concepts
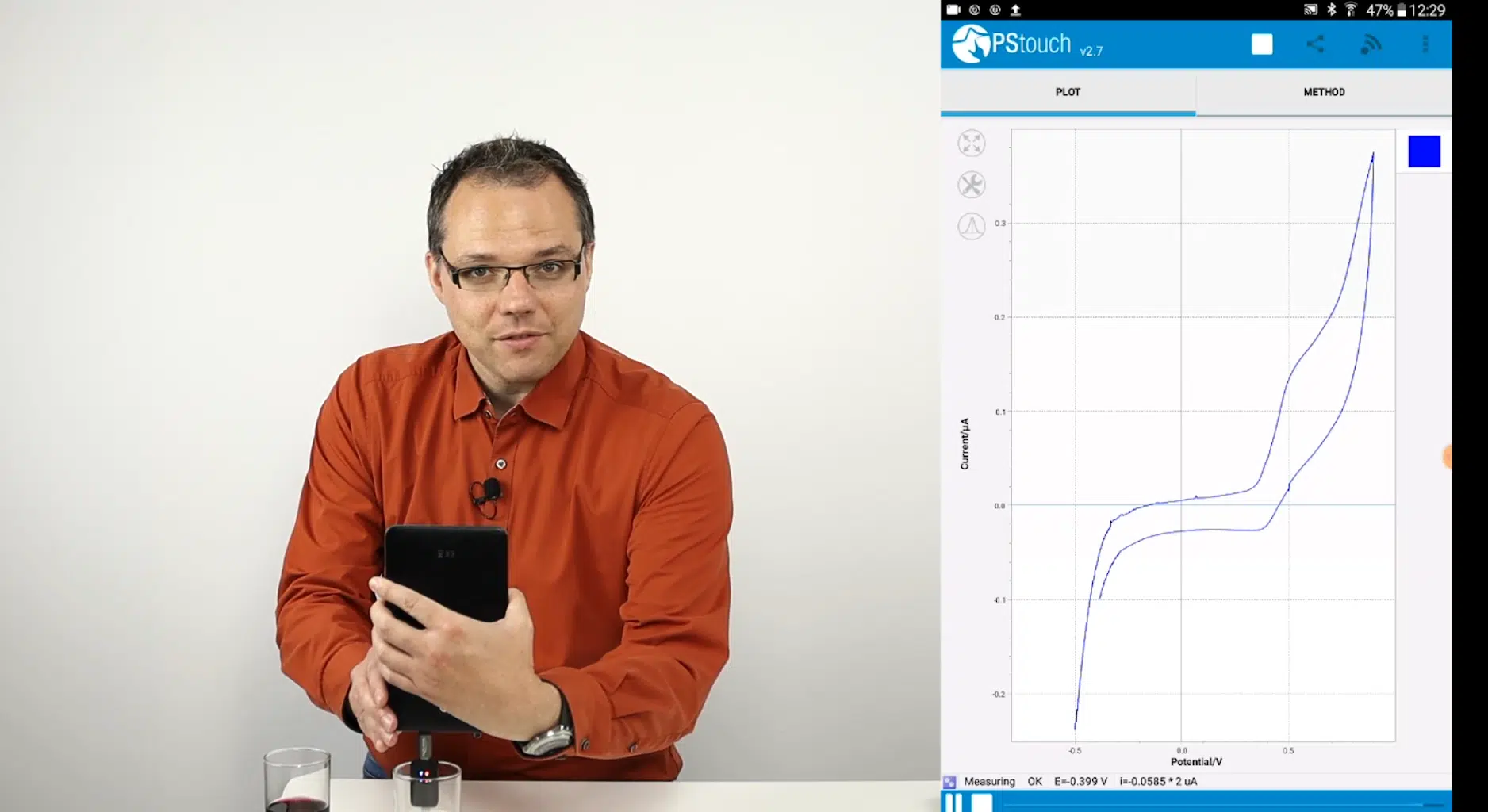
Electrochemistry in 25 minutes
In 25 minutes, you can learn what electrochemistry is, get a few example applications and learn how you can build you own electrochemical reader. After an electrochemical wine tas...

Purpose of a Sense lead
Sense leads also known as working sense leads are common for many potentiostat brands. PalmSens uses the Sense lead on all models that can supply 100 mA or more. This raises the qu...

Redox Potential
Redox potential (reduction potential of ORP) is an abbreviation for reduction and oxidation potential. A redox potential is the potential value when the reduction and oxidation of a certain metal or other chemical are in balance with each other. Redox potential is measured in Volt (V).

Potential
A potential is nothing other than the voltage or electrical voltage of a specific (liquid) substance.
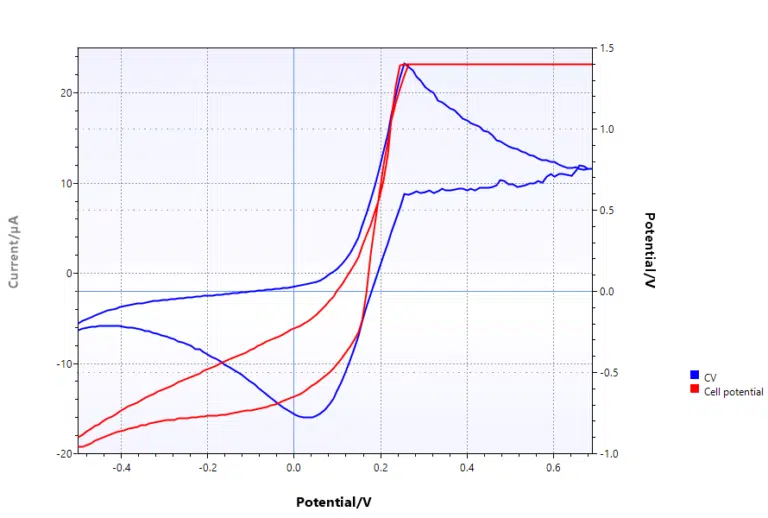
Compliance voltage
The compliance voltage is the maximum voltage that can be applied between the working and counter electrode. Another name could be the maximum cell potential.
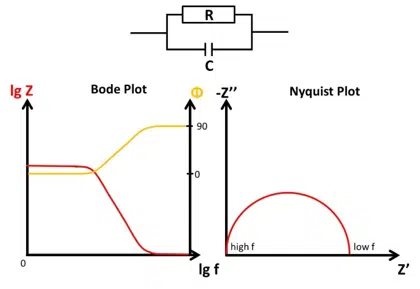
Bode and Nyquist Plot
In this chapter the two main ways of visualizing Electrochemical Impedance Spectra (EIS), the Nyquist and Bode plot, are presented and it is explained how different EIS of easy electronic circuits will be plotted in the Bode and Nyquist plot. This demonstrates the advantages and disadvantages of the two plots as well as serving as a foundation to understand the analysis of EIS by utilizing equivalent circuits.
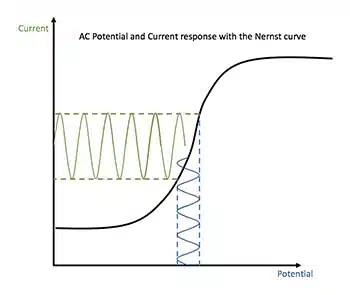
Origins of electrochemical potentials
This section is a short explanation of the processes that lead to electrochemical potential and thus to corrosion processes.

Potentiostat
A potentiostat is a device that can demonstrate the presence of a certain substance by measuring electrical current. Learn how it works
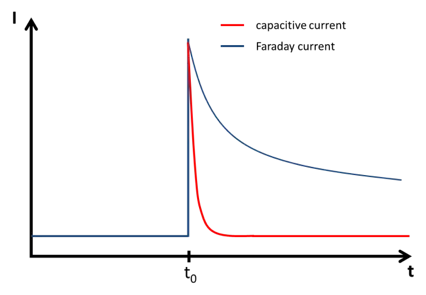
Capacitive Current
If the potential of the electrode is changed, for example during a potential step, the amount of charge the capacitor stores changes, and a current will flow that has no chemical but only a physical meaning. This is the current that charges or discharges the capacitor also known as capacitive charging current or short capacitive current.
